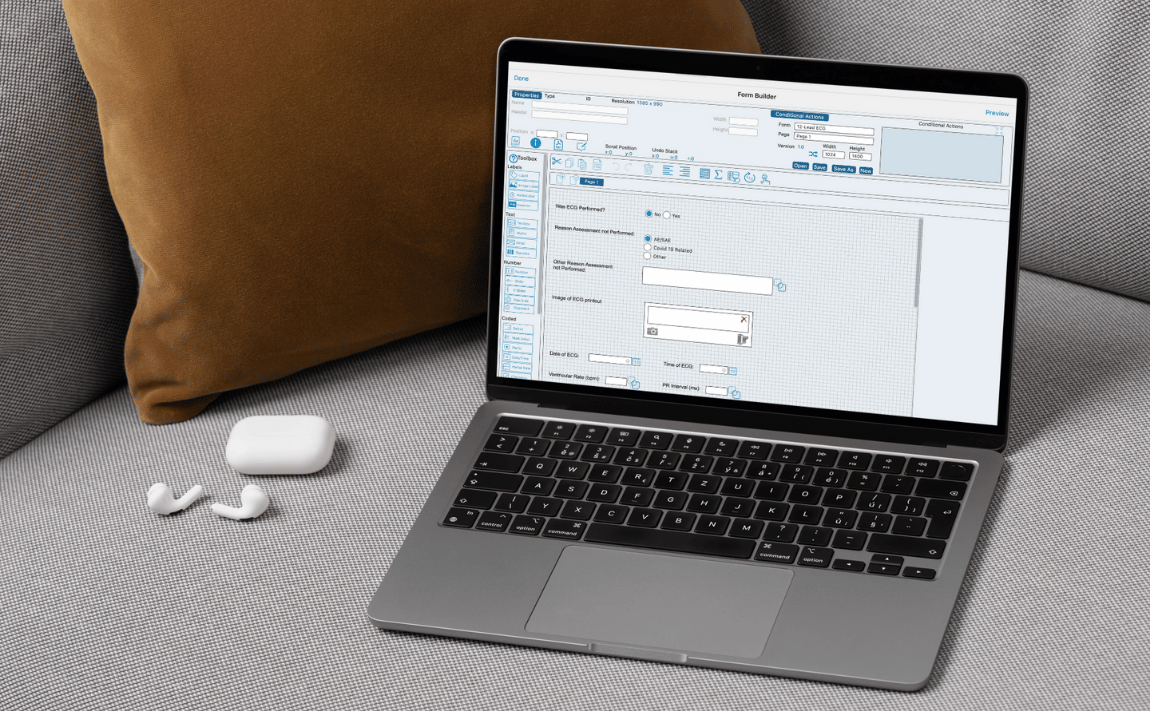
-
Crucial Data Solutions Expands TrialKit AI with Study Simulation and Validation Capabilities
RENO, NV, UNITED STATES, April 7, 2026 /EINPresswire.com/ — Crucial Data Solutions (CDS), provider of the unified eClinical platform TrialKit,
-
Crucial Data Solutions and PetPace Form Strategic Alliance to Bring Real-Time, At-Home Animal Health Monitoring to Clinical Research
BURLINGTON, Mass. & RENO, Nev.–(BUSINESS WIRE)–PetPace, the leading provider of smart wearable health monitoring collars for animals and Crucial Data
-
Crucial Data Solutions Names Dr. Leigh J. Mack Chief Medical Officer Amid Industry AI Shift
RENO, NV, UNITED STATES, July 17, 2025 — Crucial Data Solutions (CDS), provider of the industry’s only end-to-end eClinical platform
-
Crucial Data Solutions Sets Another Industry Benchmark: TrialKit Now Available as a Native macOS App
The only eClinical platform to offer native web, mobile (iOS/Android), and Mac applications, redefining the standard for modern research software.
-
Crucial Data Solutions Launches Industry-First Subscription Model to Modernize Clinical Trial Contracting and Pricing
New model empowers CROs to grow revenue, reduces costs for sponsors, and accelerates time to market RENO, NV, UNITED STATES,
-
Crucial Data Solutions Begins 2025 with AI Breakthroughs and Leadership Momentum
RENO, NV, UNITED STATES, January 8, 2025 /EINPresswire.com/ — Crucial Data Solutions (CDS), a leader in eClinical technology and a
-
TrialKit AI: Revolutionizing Clinical Research Analytics with AI
December 9, 2024 — Crucial Data Solutions is proud to announce that our groundbreaking innovation, TrialKit AI, has been recognized with
-
Crucial Data Solutions Unveils Groundbreaking AI-Driven Reporting and Analytics Module with TrialKit AI
RENO, NV, September 04, 2024 / Biotech Newswire / — Crucial Data Solutions, a leading provider of data collection and trial
-
Crucial Data Solutions Introduces TrialKit PACS to Streamline Image Management in Clinical Trials
RENO, Nev.–(BUSINESS WIRE)–Crucial Data Solutions (CDS), a leading provider of data collection and trial management software to advance clinical research,
-
Crucial Data Solutions Recognized as a Major Contender in Everest Group’s Decentralized Clinical Trial Platforms PEAK Matrix® Assessment 2023
Reno, NV, December 18, 2023 – Crucial Data Solutions (CDS), the only SaaS platform provider to empower customer-driven global clinical